Most modern ventilators actively force air into the lungs. In contrast, several recent devices change the air pressure around the body to make the chest expand and contract passively. This gentler form of ventilation is more like natural breathing. These new devices are reviving the principle of the iron lung, which saved thousands of lives during the polio epidemics of the 20th century.
From a patient perspective, assisted-breathing technologies can be traumatic. Intubation can damage the mouth, throat, or lungs even as it helps people survive. Breathing devices can also trigger profound mental distress and delirium.
Doctors and nurses are also at risk. Breathing devices expose health care workers to aerosolized pathogens. During the COVID-19 pandemic, many hospitals suffered more from staff shortages—a human crisis—than from a lack of equipment—a technology crisis.
Content from the exhibition Design and Healing: Creative Responses to Epidemics, curated by MASS Design Group and Cooper Hewitt, Smithsonian Design Museum
+++++++++++++++++++++++++++++++++++++++++++++
Iron Lung Ward, 1955
The first iron lung was used in 1928 to treat a child paralyzed by polio. An airtight chamber surrounding the body replicates the alternating pressurization and depressurization of the lungs that occurs with normal breathing. After the introduction of the polio vaccine in 1955, iron lungs became uncommon.

Photograph: Hynes Memorial Hospital, Boston, 1955, United Press Telephoto
+++++++++++++++++++++++++++++++++++++++++++++
Positive-Pressure Ventilation, 1952
The first large-scale use of positive-pressure ventilation occurred in Denmark after a polio outbreak in 1952. Medical, dental, and nursing students worked around the clock to force air into patients’ lungs with hand-squeezed rubber bags, enabling people to breathe for hours and days on end. Many of the patients were children.
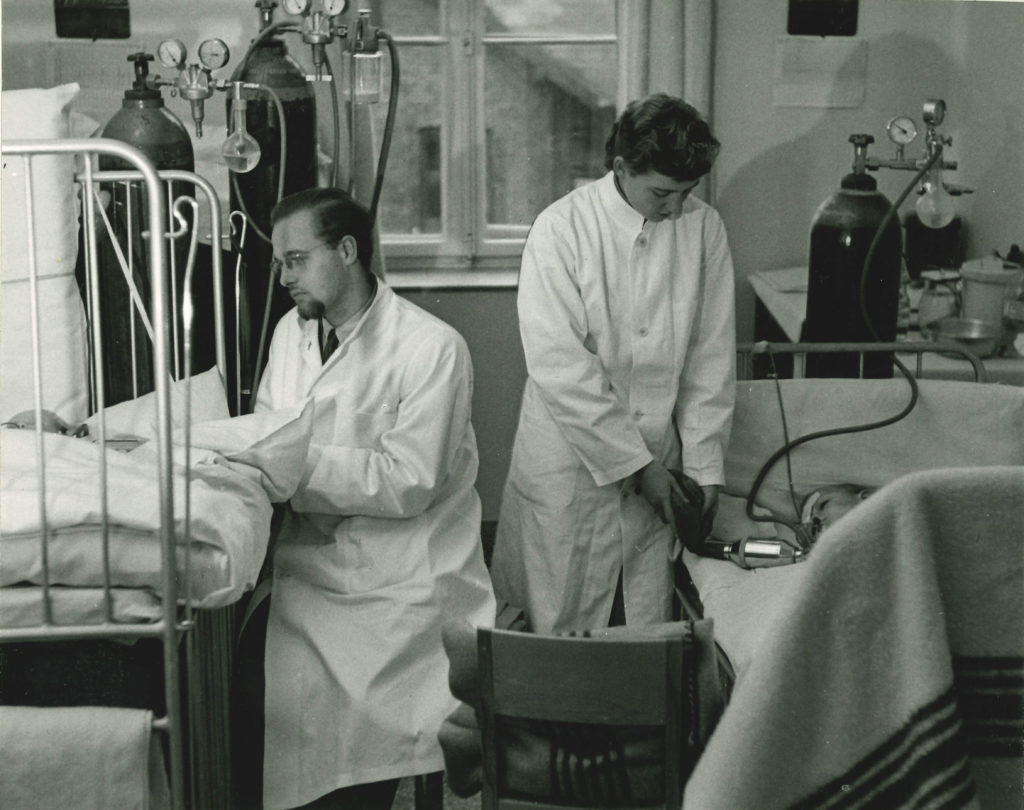
Photograph: Medical Museion, University of Copenhagen
+++++++++++++++++++++++++++++++++++++++++++++
Rescue Ventilation, 2020
To prepare for a potentially catastrophic ventilator shortage, a team of doctors and designers created a training video in March 2020 showing how to perform continuous manual ventilation. An open-source, 3D-printed adapter connects a second HEPA filter to an existing bag-valve-mask (BVM), preventing the spread of the virus. A corps of volunteer nursing and medical students could bridge the gap until a mechanical ventilator becomes available.
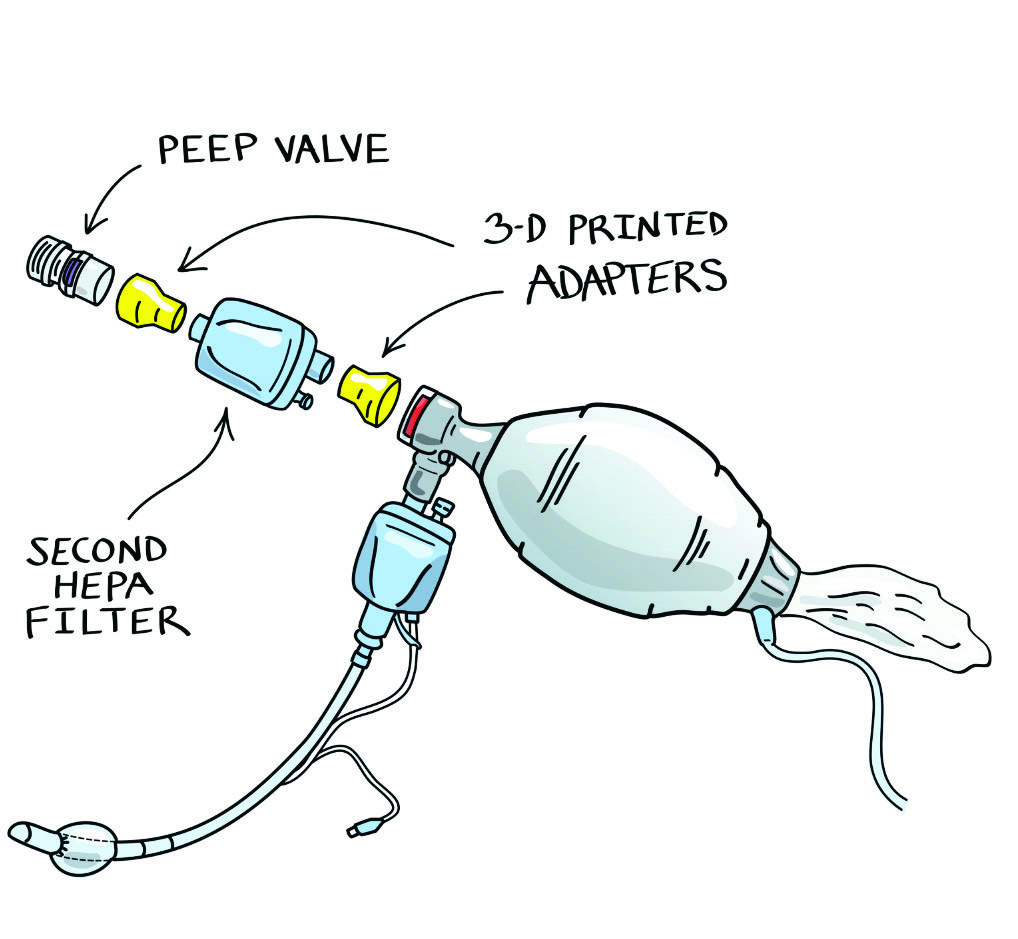
Design: Health Design Lab, FKB, and James DuCanto, MD
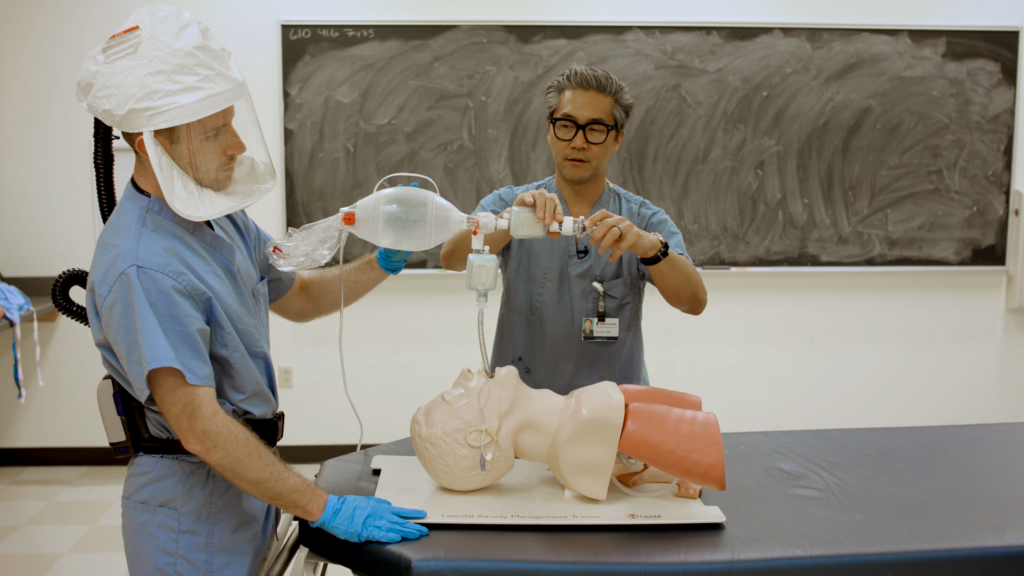
Demonstrated by Robert Ries and Bon Ku, MD; Photograph: Elizabeth Hasier
+++++++++++++++++++++++++++++++++++++++++++++
Iron Lung, 1961
A patient using an iron lung can eat, drink, talk, and read. In contrast, a patient connected to positive-pressure ventilation must be heavily sedated in order to endure treatment.

Photograph: Onan
+++++++++++++++++++++++++++++++++++++++++++++
Exovent Negative-Pressure Ventilator, 2021
Exovent, founded in the United Kingdom in 2020, is a volunteer organization devoted to designing, testing, and manufacturing a modern iron lung. Exovent does not require intubation, and the patient can remain awake. Unlike an iron lung, the device encloses only the torso area.

Photograph: Exovent, in collaboration with Marshall Aviation and Aerospace and Portsmouth Aviation
+++++++++++++++++++++++++++++++++++++++++++++
Shaash Negative-Pressure Ventilator, 2021
This working prototype was engineered and manufactured in Bangladesh, where 125,000 people die each year from COPD, asthma, and other illnesses caused by air pollution. To date, 1,500 ventilators exist for a population of more than 160,000,000. To be useful in Bangladesh, Shaash must be sturdy, affordable, transportable, resistant to dust, and easy to operate and maintain. A conventional imported ventilator costs $25,000–$50,000. Shaash will cost less than $1,000. A product successful in Bangladesh could transform health care in South Asia and Africa.
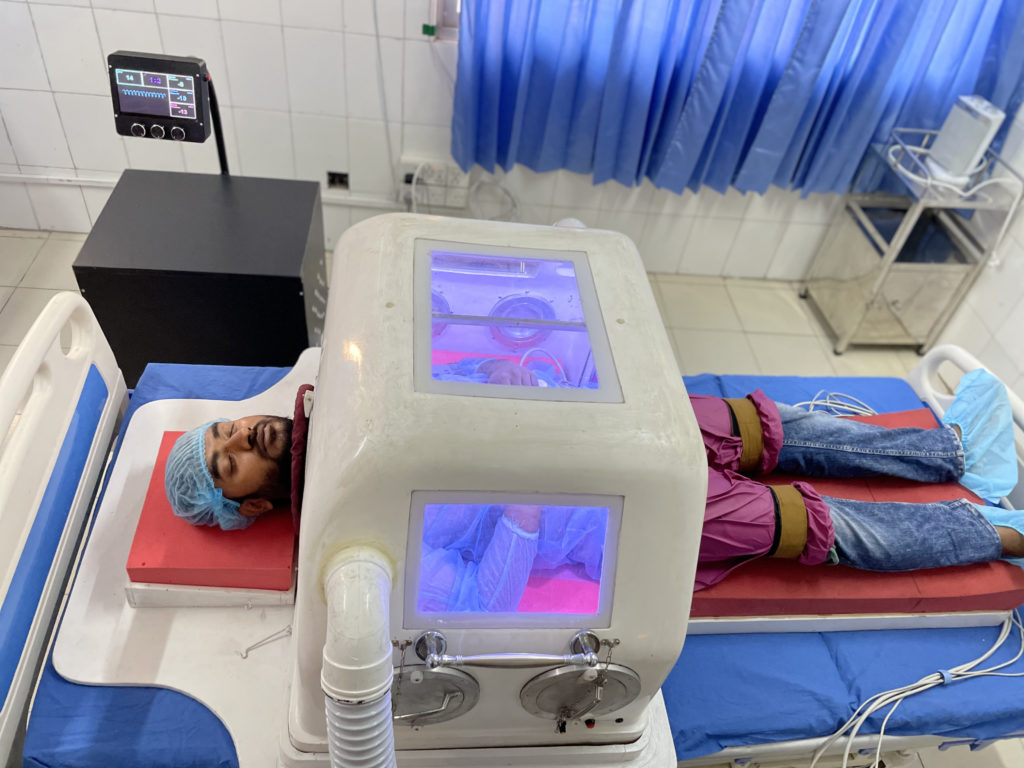
Karnaphuli Industries Limited (Chittagong, Bangladesh, founded 1963); Fiberglass, electronic components; Courtesy of Karnaphuli Industries Limited; Photograph: © Karnaphuli Industries Limited
+++++++++++++++++++++++++++++++++++++++++++++
Charlotte Valve and Mask, 2020
Cristian Fracassi and Alessandro Romaioli are Italian engineers. Dr. Renato Favero asked them to create an open-source, 3D-printed valve to connect an air supply and filtration duct to the top of a snorkel mask, hacking the mask to create an assisted-breathing device. To date, 186,000 of these devices have been used around the world.

Cristian Fracassi (Italian, b. 1983) and Alessandro Romaioli (Italian, b. 1991), Isinnova (Brescia, Italy, founded 2014); 3D-printed plastic, scuba mask; Courtesy of Cristian Fracassi
+++++++++++++++++++++++++++++++++++++++++++++
AIRA Ventilator, 2020
Robotics engineer Tyler Mantel founded The Ventilator Project (TVP) to create AIRA, an emergency ventilator that can be stored compactly and mobilized quickly in regions lacking medical stockpiles, from the rural USA to middle-income nations. The initial prototype for AIRA uses a bee bellows to move air without an external source, enabling the device to be used outside a hospital setting. The FDA approved AIRA for emergency use in March 2021.

Ventilator; The Ventilator Project team, led by Tyler Mantel (American, b. 1989); The Ventilator Project (Boston, Massachusetts, USA, founded 2020); Metal, plastic, electronics; Courtesy of TVP Health

Prototype; The Ventilator Project team, led by Tyler Mantel (American, b. 1989); The Ventilator Project (Boston, Massachusetts, USA, founded 2020); Metal, plastic, electronics; Courtesy of TVP Health

Bee bellows; Courtesy of MannLake